Translate this page into:
Immunofluorescence in Oral Dermatological Disorders- No Shiny Matter
-
Received: ,
Accepted: ,
This article was originally published by Informatics Publishing and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Dermatological diseases with underlying immunological pathosis exhibiting similar clinical manifestations have challenged even the veteran dermatologists in rendering diagnosis clinically, necessitating study of the skin lesions with immunofluroscence. The technique is similar to immunochemistry where instead of enzymes antibodies are labelled with a fluorescent dye4. It probes the study of the cell and molecular biology. Immunofluorescence permit early diagnosis, treatment and subsequent monitoring of disease activity in patient with potentially life threatening disease. It is widely used in the field of vesiculo-bullous lesions and other related oral dermatological disorders by demonstrating antibodies either in biopsy tissue or in the serum separated from blood collected from the patients.
Keywords
Immunofluorescence
Pemphigus
Pemphigoid
Epidermolysis Bullosa
Mucous
Membrane Pemphigoid
Lichen Planus
1. Introduction
Immunofluorescence is an investigation used to diagnose patients with dermatological disorders[1]. It is a staining technique used to demonstrate the presence of antibodies bound to antigens in tissues or circulating antibodies especially when the clinical and microscopic findings are inconclusive. This test is carried out by incubating a biopsy specimen with a fluorescein – conjugated anti globulin. As a result of, the unattached antibodies are washed off and the suspected fluorescence labelled conjugate is applied[2,3]. The unbound conjugates are washed off and the slides are viewed under fluoresecent microscope. Positive sample indicates apple green fluorescence[4].
2. Physics of Immunofluorescence
Certain elements, fluorochromes, bear valence electrons in the outer shell that absorb energy and shift to the inner shell drawing closer to the nucleus by which the element loses its stability. In order to return to its natural or stable state, these electrons return to the outer shell by emitting the absorbed energy in the form of low wavelength radiation. It is this principle that is utilized in immunofluorescence.
In immunofluorescence technique, the synthesized antibodies are complexed with fluorochromes that emit low wavelength radiation, which is spontaneous, that can be visualized by an ultraviolet microscope. Currently, the fluorochromes utilized are fluorescein isothiocyanate which produce an apple green colour and tetramethylrhodamine isothiocyanate producing red colour fluroscence.
For studying tissue specimens derived from patients with immunological disorders, the synthesised fluorescent antibody detects the human antibody forming an antibody complex (Figure 1).
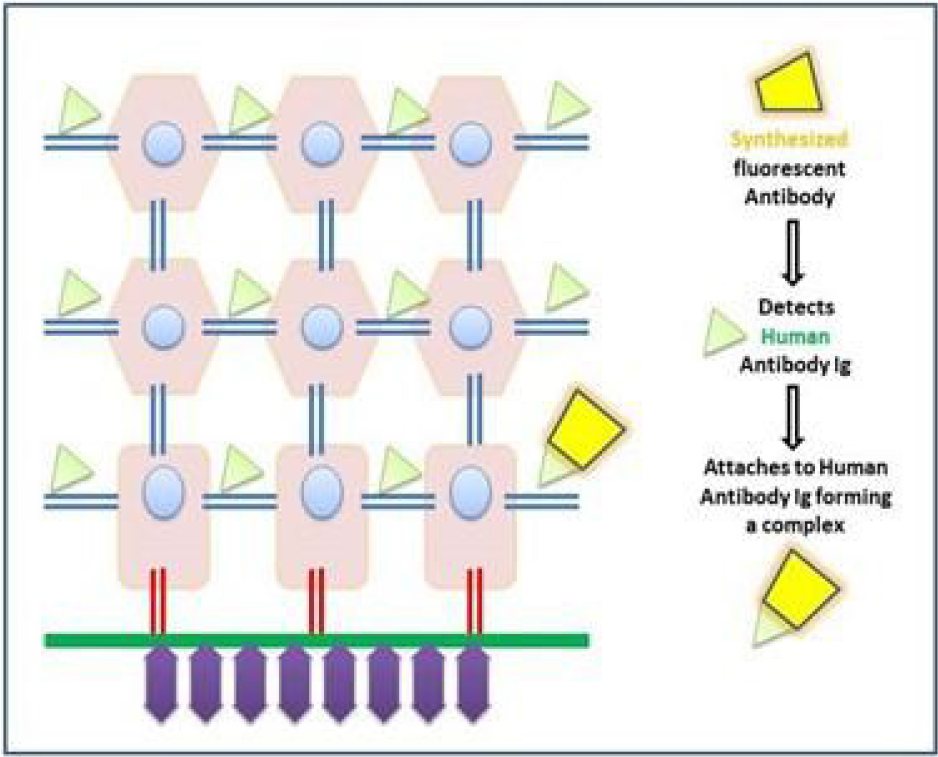
- Application of fluorescent antibody on tissue specimen.
When the high energy short wavelength light from microscope falls on the synthesised fluorescent antibody, it will emit light of lower energy with longer wavelength which can be visualized using a fluorescent microscope (Figure 2).
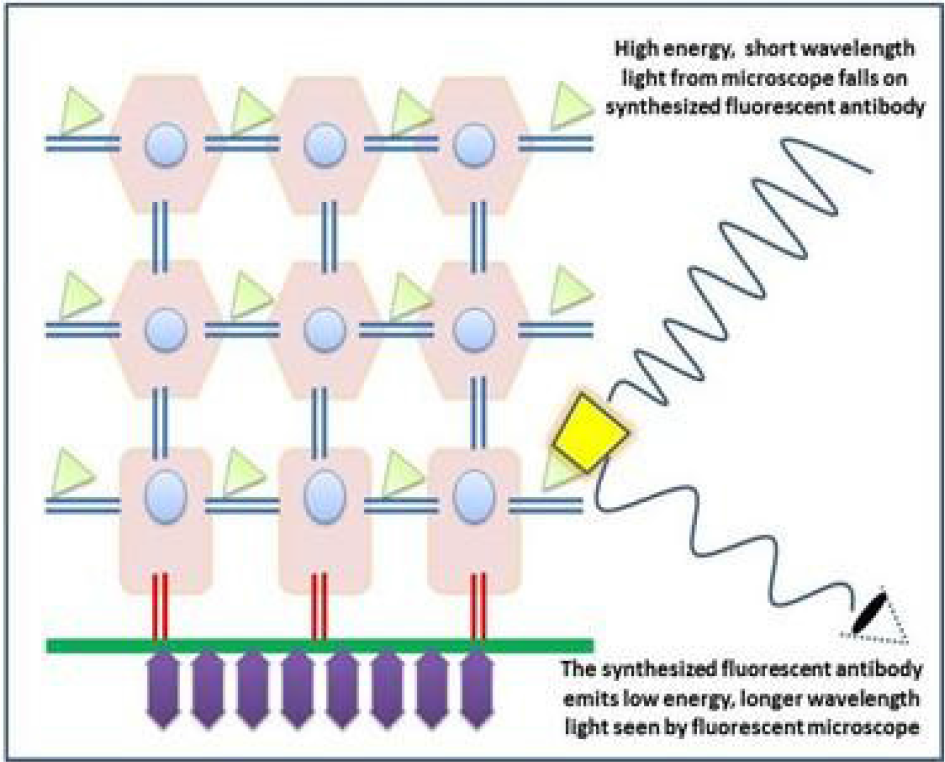
- Visualization using fluorescent microscope.
3. Application in Pathology
In normal epithelium, the basal cell layer comprise of short columnar cells that are attached to the adjacent epithelial cells by desmosomes, while the cells of the basal layer are attached to the basement membrane by hemidesmosomes. The basement membrane is attached to the underlying connective tissue by anchoring fibrils (Figure 3).
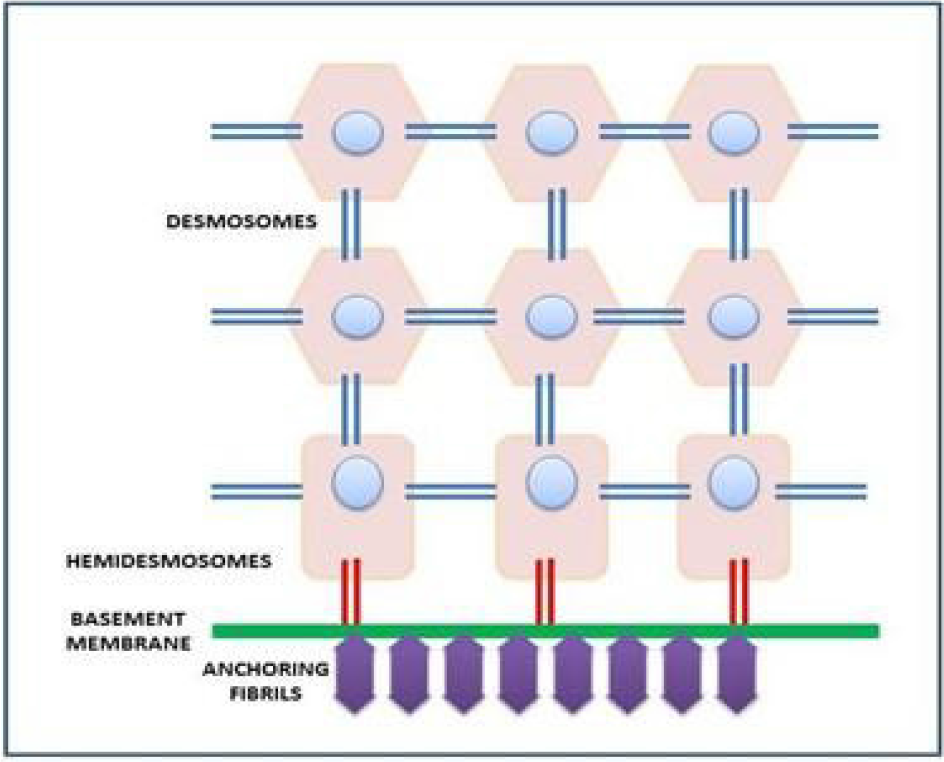
- Normal epithelium with attachment.
3.1 Pemphigus
Antibodies (IgG) attach to the desmosomal component of the suprabasal layers causing weakening of the epithelial attachment leading to formation of an intraepithelial cleft (Figure 4).
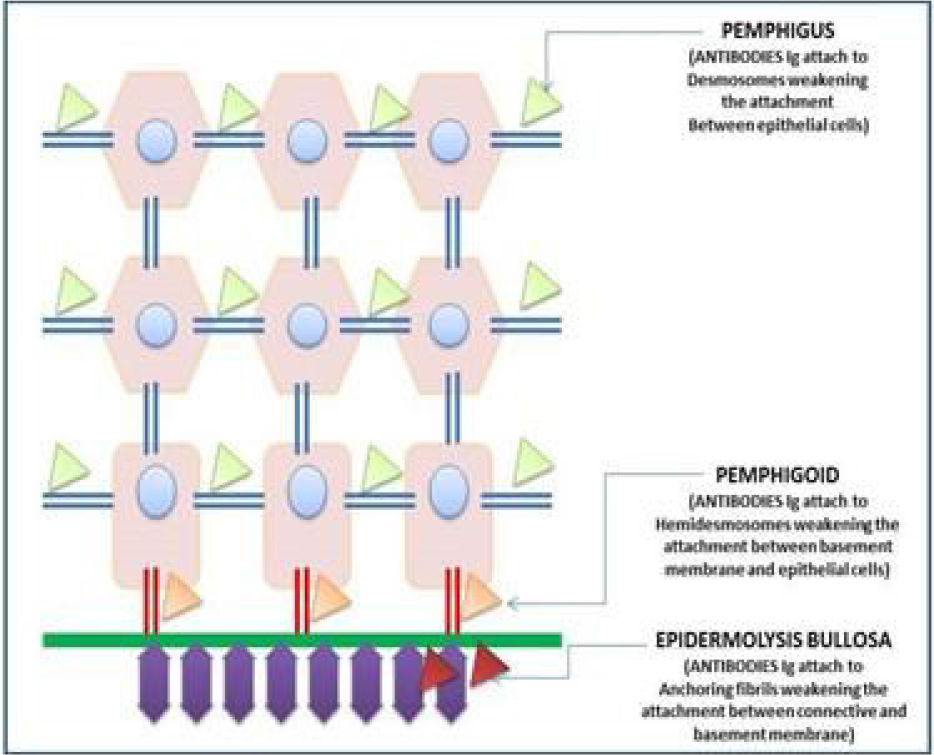
- Antibody attachment seen in Pemphigus, Pemphigoid and Epidermolysis Bullosa.
3.2 Pemphigoid
Antibodies (IgG, IgA, C3) attach to the hemidesmosomes of the basal cell layer, causing weakening between the epithelial and the basement membrane leading to a subepithelial cleft (Figure 4).
3.3 Epidermolysis Bullosa
Antibodies (IgG, IgA, IgM, C3) attach to the anchoring fibrils causing weakened attachment between the connective tissue and basement membrane which leads to formation of bullous lesion of the skin and mucosa with minimal trauma (Figure 4).
4. Types of Immunofluorescence
Direct immunofluorescence.
Indirect immunofluorescence.
Complement fixation.
Immunoelectron microscopy.
4.1 Direct Immunofluorescence
Primary or direct immunofluorescence uses a single, primary antibody, and is chemically linked to a fluorophore. In this technique antigens are detected in the tissue of the patient and the labelled antibodies are directly applied to tissue[5]. The primary antibody predominantly recognizes the IgG antigen and binds to the epitope which is in the specific region. If antigens of interest are present in the tissue it will exhibit fluorescence when viewed under the fluorescent microscope.
4.2 Indirect Immunofluoresence
Indirect immunofluorescence is a highly sensitive technique. This method has an advantage that it requires only a blood sample and is thus adopted for patients with extensive ulcers where intact oral mucosa would be difficult to obtain for biopsy. The serum separated from the patient’s blood sample is added to the substrate and is incubated resulting in fixation of circulating antibodies to the antigen in the substrate. The next step in this procedure consists of the application of fluorescein labelled antihuman gammaglobulin to the substrate.
4.3 Complement Fixation
This is a modification of indirect immunofluorescence where the patient’s serum, separated from the collected blood sample, is smeared on a substrate and a specific complement is added. The synthesized fluorescein anti-complement antibodies are used to detect the presence of complement in the tissue[5].
4.4 Immunoelectron Microscopy
In this technique, the tissue sample is “immune-stained” by using antibodies complexed with horse radish enzyme labelled with heavy metal such as colloidal gold to localize the antigens providing ultrastructural visualization. It is analogous to direct immunofluorescence technique[5]. This technique is helpful in differential diagnosis of heredi-tary Epidermolysis Bullosa, where antigen mapping is not significant.
5. Antigens in Oral Dermatological Disorders
5.1 Pemphigus Vulgaris
Pemphigus vulgaris is one of the commonest clinical forms of the autoimmune vesiculobullous disorder, Pemphigus. Direct Immunofluorescence of perilesional skin in pemphigus exhibit linear or granular pattern of deposition of immunoglobulins and C3 in the epidermal intercellular spaces[2]. It reveals uniform “fish like net pattern” or “chicken wire pattern” (Figure2).
5.2 Bullous Pemphigoid
Bullous pemphigoid is another immunological dermatological disorder where, as mentioned in above, exhibit linear, wavy, tubular and granular like pattern between the basal layer and the underlying basement membrane. Clinically, this disorder presents with ocular symptoms prior to oral manifestations. Since this disorder shows histologically a subepithelial cleft, clinically, the roof of the vesicles or bullae comprising of full thickness of epithelium thereby rupture late compared to Pemphigus1 (Figure 4).
5.3 Gestational Pemphigoid
This can be seen during or soon after pregnancy. In DIF, it typically demonstrates linear C3 and IgG may be seen in 30% cases.
5.4 Lichen Planus
Clinically, erosive Lichen planus can present as eroded areas mimicking other dermatological disorders such erosive mucous pemphigoid and can be distinguished by direct immunofluorescence[6]. Lichen planus reveals a characteristic pattern of fibrinogen deposition along the basement membrane and spreading irregularly into the superficial lamina propria, which has been described as “shaggy or fibrillary pattern”. The most frequently present immune deposits are IgM and fibrinogen.
5.5 Epidermolysis Bullosa Acquisita
This disease is an acquired subepidermal blistering disease characterised by the presence of autoantibodies to type VII collagen in basement membrane zone. In epidermolysis bullosa acquisita, intense IgG deposition is present[2]. The intensity of C3 deposition is usually less than that of IgG (Figure 4).
5.6 Lupus Erythematous
The application of direct immunofluorescence reveals various subsets of the lupus erythematous according to its morphology and frequency of deposition[6]. Direct immunofluorescence reaction in specimen of oral mucosal LE exhibits coarse granular deposits of C3, IgM, IgA and IgG in the basement zone[7]. This reaction pattern can be seen in other disorders such as mixed connective tissue diseases such as dermatomyositis, scleroderma, drug eruptions, and vasculitis.
5.7 Mucous Membrane Pemphigoid
Mucous Membrane Pemphigoid (MMP) exhibit a linear pattern of immunofluorescence at the basement membrane, detecting deposition of all three antibodies, anti IgG, anti IgA, and anti-C3 antibodies[8] (Table 1).
| Lesion | Histopathological Features | Immunofluorescence | |
|---|---|---|---|
| Antibodies | Pattern | ||
| Pemphigus vulgaris | Intraepithelial clefting | IgG | Net like distribution |
| Mucous membrane pemphigoid | Subepithelial clefting | IgG, IgA, C3 | Linear distribution along basement membrane |
| Lichen planus | Hyperkeratosis Saw toothed rete ridges, band like infiltrates of lymphocytes | IgG, IgA, C3 | Shaggy or fibrillar pattern |
| Lupus erythematous | Hyperkeratosis, follicular plugging(keratin packed in opening of hair follicle) | C3, IgM, IgA, IgG | Coarse granular deposition |
| Epidermolysis bullosa | Intact stratum corneum with vesicle formation in the lower epidermis at the basal layer | IgG, IgA, IgM, C3 | Multiple deposits at the basement membrane zone |
6. Significance in Dentistry
In the differential diagnosis of oral ulcers from dermatalogical disorders in cases with presentation of single or isolated ulcers.
Direct immunofluorescence is used in the oral dermatological disorders mainly in oral bullous disease and lupus erythematosus.
Indirect immunofluorescence applied in pemphigus vulgaris, paraneoplastic pemphigus.
Immunofluorescence is used to locate the soluble dental plaque components in human gingiva affected by periodontitis.
It locates the Streptococcus mutans in the dentinal tubules of open infected root canals.
Sensitive method in detecting Porphyromonas gingivalis by polymerase chain reaction and its possible clinical application.
Helps in the observation of maturation of dental plaque.
Application of antimicrobial oral fluoride varnishes in patients undergoing orthodontic treatment.
7. Conclusion
The specialized usage of indirect and direct immunofluorescence has been widely proposed in various methods of studies of oral dermatological disorders such as Pemphigus, Pemphigoid, Lichen Planus, Lupus Erythematous, and Epidermolysis Bullosa by the visualization of antigens within cells using antibodies as fluorescent. Here the advantages over shadow the disadvantages in the practice of investigative dentistry particularly when the clinical and microscopic findings are indecisive.
References
- Immunofluorescence and its oral diagnostic applications. Journal of Orofacial Science. 2013;8(5):3.
- [Google Scholar]
- Immunofluorescence in dermatology. Indian J Dermatol Venereol Leprol. 2012;78(6):677-91.
- [Google Scholar]
- Immunofluorescence in oral mucosal disease. Journal of Orofacial Science. 2013;5(1):6-10.
- [Google Scholar]
- Textbook of Oral and Maxillofacial Pathology (3rd edition). Saunders: Elsevier; 2009. p. :771.
- [Google Scholar]
- Technique of immunofluroscence and its significance. Indian J Dermatol Venereol Leprol. 2008;74(4):15-9.
- [Google Scholar]






