Translate this page into:
Hard Tissue Architecture of Tooth – No Strain with Stain
*Address for correspondence:
This article was originally published by Informatics Publishing and was migrated to Scientific Scholar after the change of Publisher.
Abstract
One of the methods for visualizing and studying the hard tissues of the tooth is by preparation of ground sections of the desired area of the tooth. This method is relatively simple and enables us to appreciate the site and arrangement of various anatomical units of enamel, dentin and cementum. However, these sections are unstained and the features observed are based on the degree of light permeating through the sections. In this experiment, natural tooth were preserved in haematoxylin and eosin to study histological components of tooth. Twenty recently extracted natural teeth were utilized for this experiment and divided into two groups. In group 1, apical portion of the tooth were sealed and perforations were made on the different aspects of the tooth. In group 2, the apical portions of the root were flattened and the apex kept open. Both groups were preserved in haematoxylin and eosin to permit imbibition of the stain. Following which, ground sections were prepared and viewed under a microscope.
Keywords
Haematoxylin and Eosin Stains
Histological Components
Ground Section
1. Introduction
Enamel, dentin and cementum form the hard tissue component of the tooth and one of the methods to observe the histology of these hard tissues is by preparation of unstained ground sections which rely on the degree of light that passes through the crystal arrangement. Staining of specimens provide a contrast to delineate the boundaries and identification of components of a cell. The present study attempts to stain the structural components of the hard tissues to better appreciate the arrangement and architecture of the hard tissues of the tooth.
2. Aim
The aim of the experiment is to observe the hard tissue architecture of tooth under a stereomicroscope and compound microscopes.
3. Objectives
To study the hard tissue components of the tooth by preserving the tooth in hematoxylin and eosin stain for a period of twenty days with perforations prepared on different aspects of the crown and root portion.
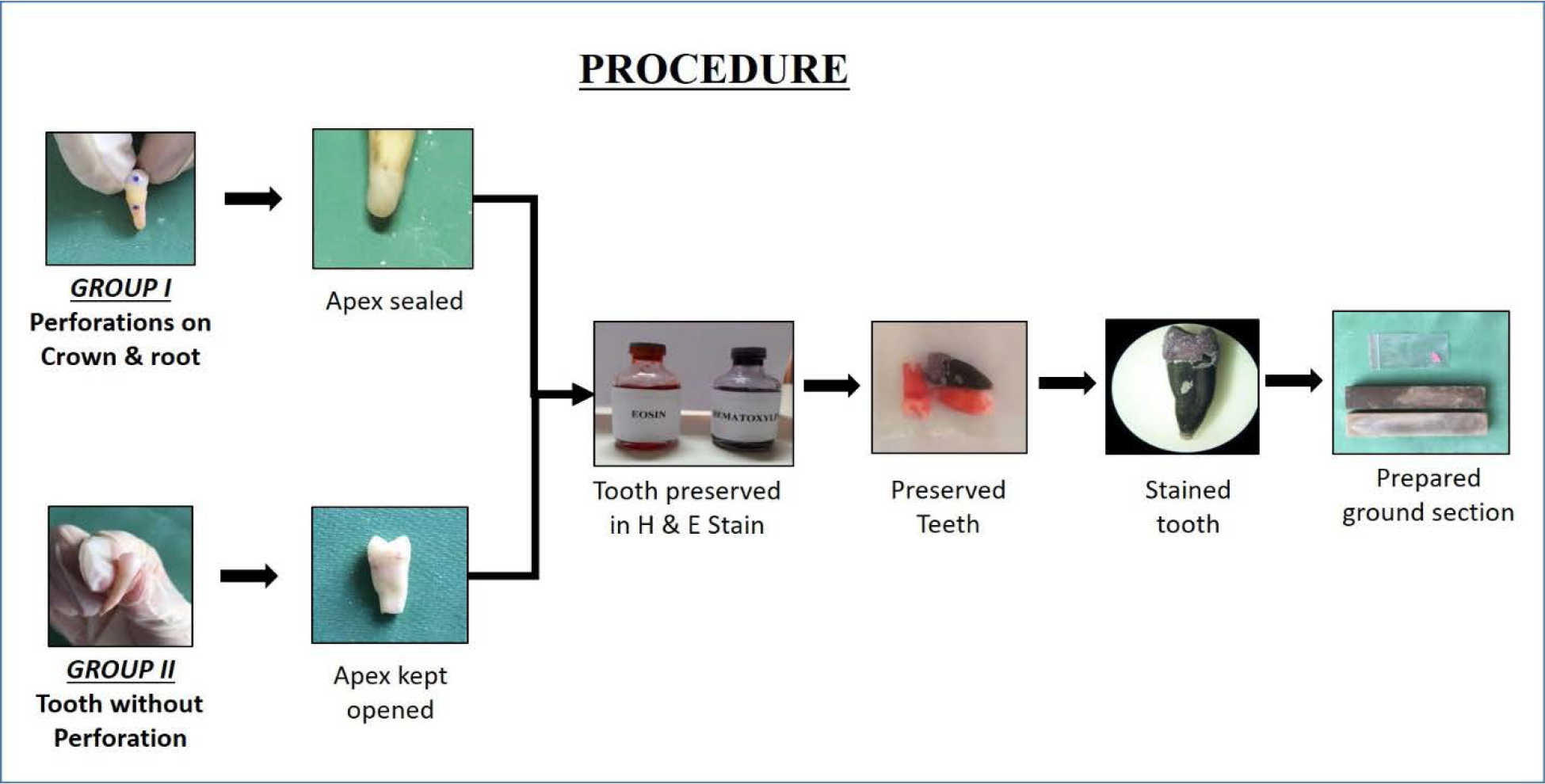
- Procedure.
4. Methodology
Recently extracted twenty natural human teeth were collected from the private clinics for this experiment. All the collected teeth were kept in hydrogen peroxide solution for 24 hours followed by a rinse in sterile water. For this experiment, the twenty teeth were divided equally into two groups, Group I and Group II.
In Group 1, the apical portion of the tooth was sealed with light cure composite resin. After which, three perforations were made, one on the buccal aspect of mid portion of crown, second on the cementoenamel junction and the third on the root portion of each tooth with the help of contra angle hand piece with diamond straight fissure bur. The depth of the perforation depends on the thickness of enamel and cementum, was 0.75 to1 mm in the crown, 0.50 mm in the cement enamel junction and 0.25 mm in the root to permeate stains through dentinal tubules.
In Group II, apical one third of tooth was flattened with the help of hand piece and straight fissure bur to keep the apex open and the patency of the canals were verified with endodontic files.
Both groups of the teeth were preserved in haematoxylin and eosin stain for a period 7 to 20 days for imbibition of stain through the perforations.
Teeth from each group were taken out from the stain on 5th, 10th and 20th day and were seen under the stereo-microscope before sectioning followed by compound microscope after sectioning. The ground sections were prepared in two steps; in first step, the sections were prepared using a laboratory lathe with wheel until the tooth reaches 4 mm thickness. In step two, using a flat carborundum stone with pumice and water. Grinding slabs has various grades, first with flat, rough carborudum stone till a section of 2 to 3 mm was obtained and then static carborundum stone until it reaches 1 mm thickness followed by fine carborundum stone till the section of 30-40 microns thickness was obtained then the sections were allowed to dry. Finally cleaned and dried prepared longitudinal ground section was mounted on slide using DPX solution with coverslip and viewed under compound microscope.

- Eosin stained Incremental lines of Retzius were clearly seen at cervical region of tooth.
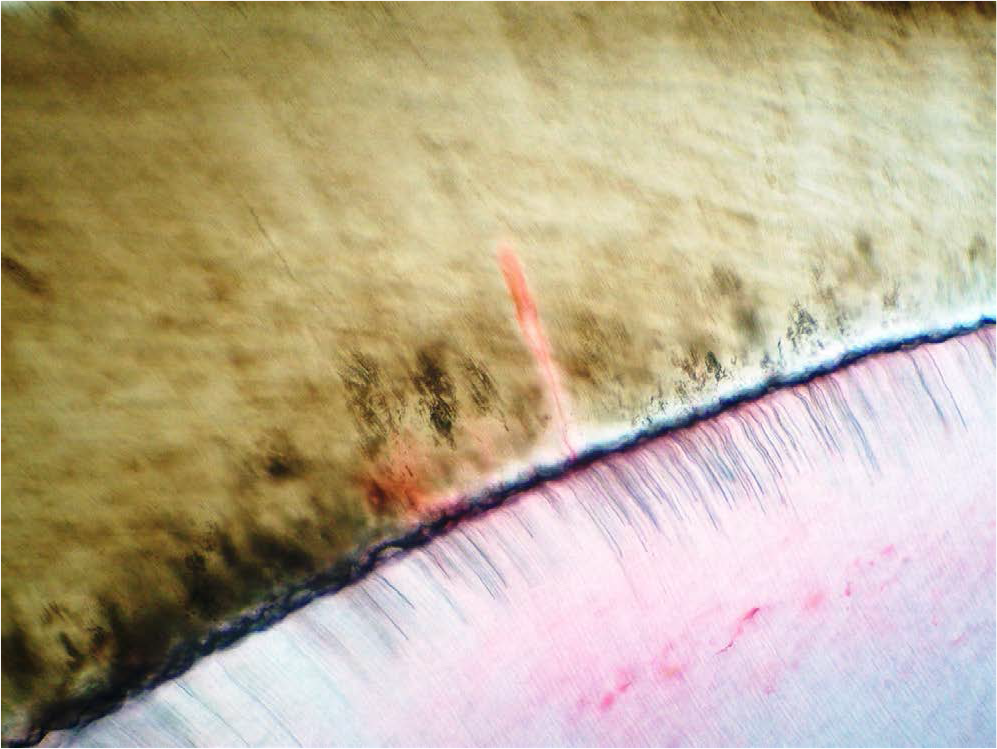
- Enamel spindles were stained by eosin.

- Eosin stained Interglobular dentin seen beneath dentinoenamel junction under transmitted light.

- High magnification of globular masses were seen near cirumpulpal region of dentin.

- Eosin stained Individual odontoblastic process.
5. Inference
The teeth preserved in haematoxylin did not take up stain.
The tooth with open root apex (Group II) did not take up the stain as effortlessly as teeth which had perfo-rations made on the crown, cementoenamel junction and root portion (Group 1).
6. Discussion
The microscopic anatomy of the tooth have always been an intriguing and difficult aspect to visualize under a compound microscope as the integration of hard and soft tissues are distorted following decalcification and histo-logical processing. Enamel being highly calcified with almost 96% inorganic component, is studied by preparation of extremely thin sections of tooth which do not take up any stain owing to its significant inorganic content and may easily fracture during reduction. Dentin has comparable calcification or inorganic component of bone with the difference in the presence of odontoblastic process (soft tissue) lined by inorganic ions forming tubular structure extending from the periphery of pulp to the junction of enamel. This arrangement is examined by preparation of ground sections to observe the inorganic component and decalcification to study the odontoblastic process. Cementum calcification varies along the length of the root with acellular cementum in the cervical portion and cellular cementum in the apical portion. Since ground sections is one of the methods employed to study these features, an attempt was made to stain these hard tissues with routinely used haematoxylin and eosin stain to better appreciate the integral arrangement of hard and soft tissues.
The features observed in the longitudinal ground sections are given below in the order of enamel, dentin and cementum.
6.1 Enamel
Eosin stained incremental lines of Retzius were well appreciated at the cervical portion than the other region of the tooth.
Enamel spindles also exhibited staining by eosin

- Terminal branches of dentinal tubules were seen near cementodentinal junction. Branches were anastomosing with adjacent tubules.
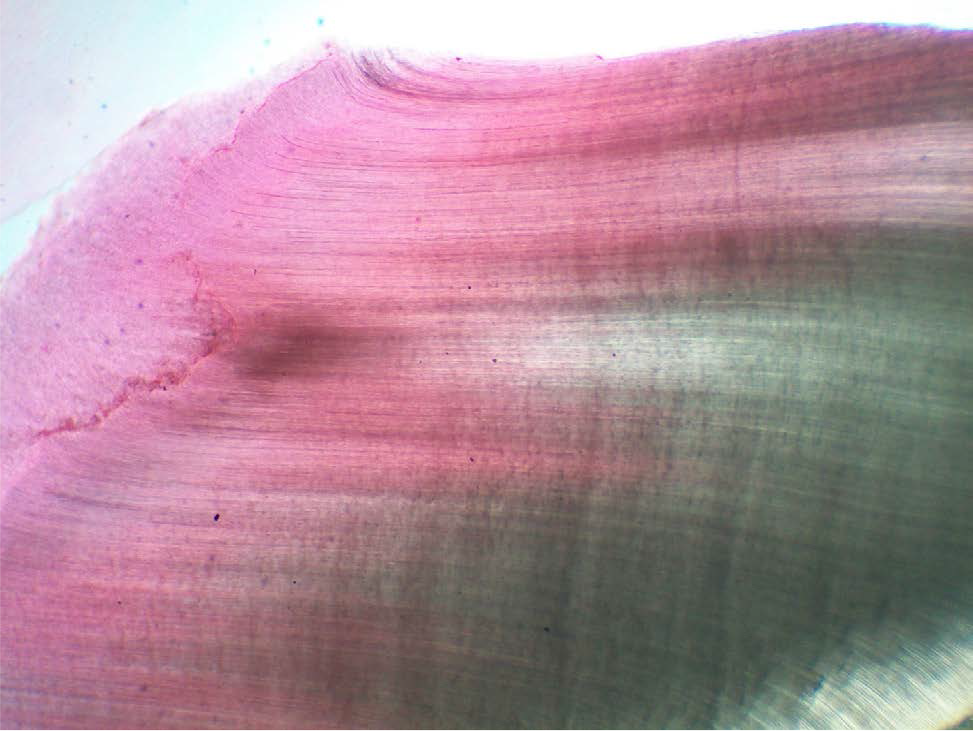
- S-shaped dentinal tubules were well observed on crown from peripheral region of pulp towards dentinoenamel junction in longitudinal sections.
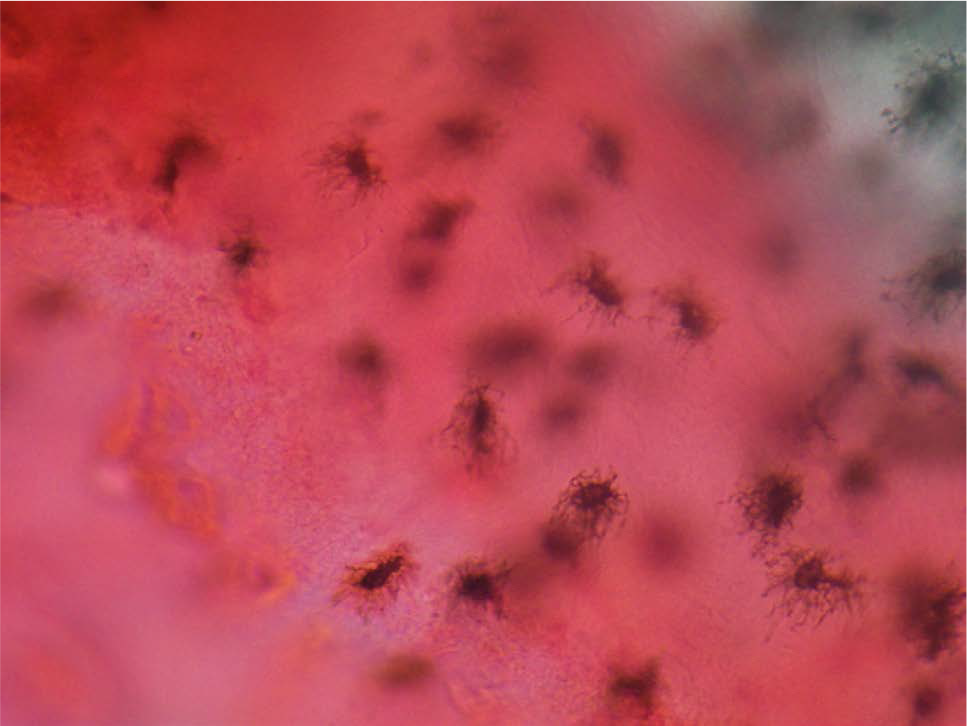
- Cellular cementum was stained by eosin which provides great contrast to cementocytes. Note the canaliculi of cementocytes were anastomosing with adjacent one and also canaliculies were prominent towards periodontal ligament.
6.2 Dentin
Eosin stained S-shaped dentinal tubules were clearly seen at crown portion of the tooth.
Inter globular dentin at root portion.
Globular masses were seen at the circumpulpal region stained by eosin.
Inter connections between terminal branches of odontoblastic processes were well appreciated because of eosin stain.
Longitudinal sections of individual dentinal tubules were stained by eosin.
6.3 Cementum
Cementocytes of cellular cementum were appreciated clearly because of contrast provided by the eosin stain.
7. Conclusion
Ground section of tooth is one of the methods to study anatomy of tooth under a microscope. The fact still remains that these unstained sections provide an obscure image of the histology and arrangement of the hard tissues of tooth. Staining provides contrast and depth to the observer to comprehend the integrity and organization of hard and soft tissue in a two dimensional plane. The purpose of the experiment was an attempt to study the microscopic anatomy and architecture of the tooth by employing routinely used haematoxylin and eosin stain. Among the two stains utilized, eosin stain was taken up by the dentin portion, cervical portion of enamel and cementum while haematoxylin was not taken up by the hard tissues. Staining did advance the histological features when compared with unstained ground sections and application of other stains has to be investigated to view the hard and soft tissues of tooth.
References
- Orban’s Textbook of Oral Histology (12th ed). Elsevier publications; Mosby; 2010. p. :95.
- [Google Scholar]
- Few tips for making ground sections of tooth for research purpose: A review. Journal of Punjab Academy of Forensic Medicine and Toxicology. 2006;6:11-3.
- [Google Scholar]
- Histological preparation of teeth and tooth growth. A review. Archives of Oral and Dental Research. 2015;2:1-5.
- [Google Scholar]






